Charlotte K Barton1, Kathryn Seabaugh 1, Ben Gadomski 2, Kevin Labus 2, Ben Holmes 3, Nathan Castro 3, Mike Hawes 4, Brad B Nelson 1, Laurie R Goodrich 1
1 Orthopaedic Research Center, Translational Medicine Institute, College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, CO. 2 Department of Biomedical Engineering, Colorado State University, Fort Collins CO. 3 Nanochon Inc, Washington DC. 4 Charter preclinical services, Hudson, MA.
Background: The treatment of full thickness cartilage defects remains a challenge. Repair tissue is structurally and functionally inferior to healthy articular cartilage, therefore the development of a treatment that improves outcomes of these lesions is highly sought after. The study objective was to evaluate the use of Chondrograft™ compared to empty defects for the repair of full thickness cartilage lesions. The hypothesis was that Chondrograft™ treated defects would have improved repair tissue fill and integration, with improved biomechanical functional compared to empty defects.
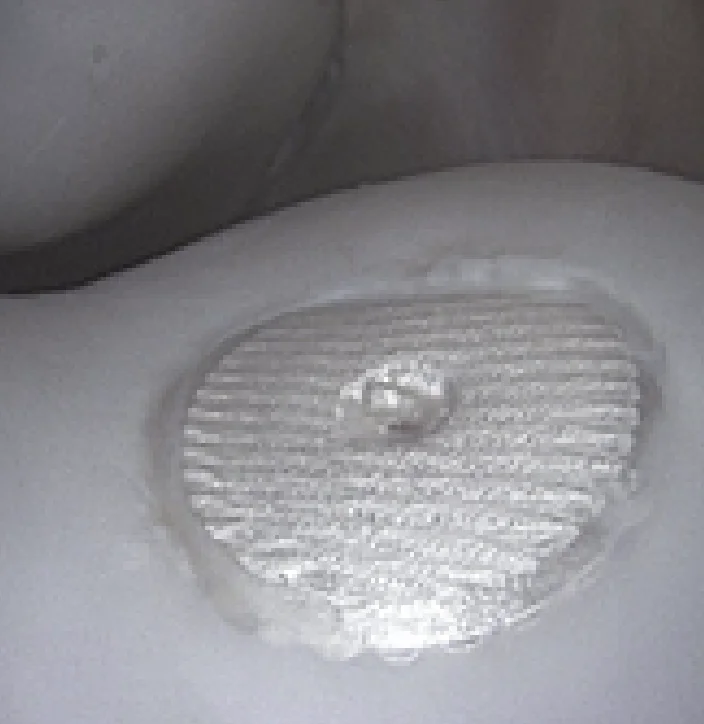
Experimental Design: The study included 12 healthy adult horses. On Day 0, bilateral stifle arthroscopy was performed, and 15mm full-thickness cartilage defects were created on the lateral trochlear ridge of each femur. One limb received the Chondrograft™ implant while the other limb served as the empty control (Figure 1). Lameness examinations (joint effusion and range of motion) evaluations occurred every 4 weeks. A second look arthroscopy with ICRS scoring was performed at 12 weeks. Endpoint testing included MRI (MOCART 2.0), arthroscopic gross assessment, histology and biomechanical indentation at 52 weeks.
Results: ICRS scores at 12 weeks were significantly improved in the Chondrograft™ treated defects vs control defects (4.08 vs 11.23) and 52-week ICRS scores significantly improved in the Chondrograft™ treated defects vs control defects (Figure 2). Clear differences were observed arthroscopically between Chondrograft™ treated defects vs control defects at week 52 as well (Figure 3). Biomechanical testing showed no significant difference in stiffness between Chondrograft™ and significantly normal cartilage however control repair tissue was weaker than normal cartilage (Figure 4). Histology of Chondrograft™ treated limbs had significantly higher ICRS (35.8 vs 25.9) and modified O’Driscoll scores (15.6 vs 12.5) compared to empty control defects (Figure 5).
Conclusions: Chondrograft™ results in excellent defect filling and integration to the surrounding cartilage, with biomechanical function similar to normal healthy articular cartilage (Figure 6). These findings suggest that Chondrograft™ is a viable option to improve outcomes of cartilage defects.
Figure 1. 15mm full thickness cartilage defects were made on the lateral trochlear ridge of the femur.
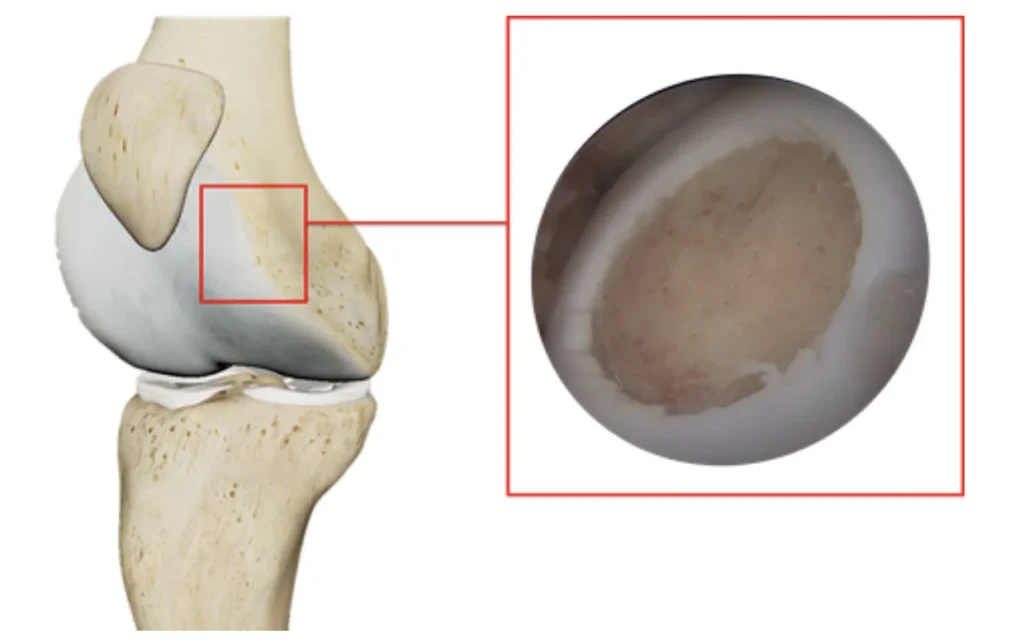
Figure 2. ICRS scores of Chondrograft™ and control defects at 52-weeks.
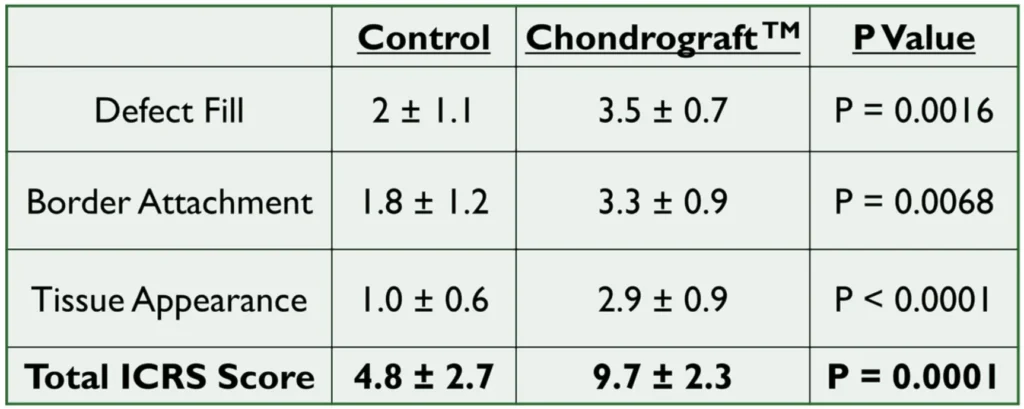
Figure 3. Arthroscopic images of a) Chondrograft™ treated limb and b) control limbs at 52-week endpoint.

Figure 4. No significant difference in stiffness between Chondrograft™ and normal cartilage. Control repair tissue significantly weaker than normal cartilage.
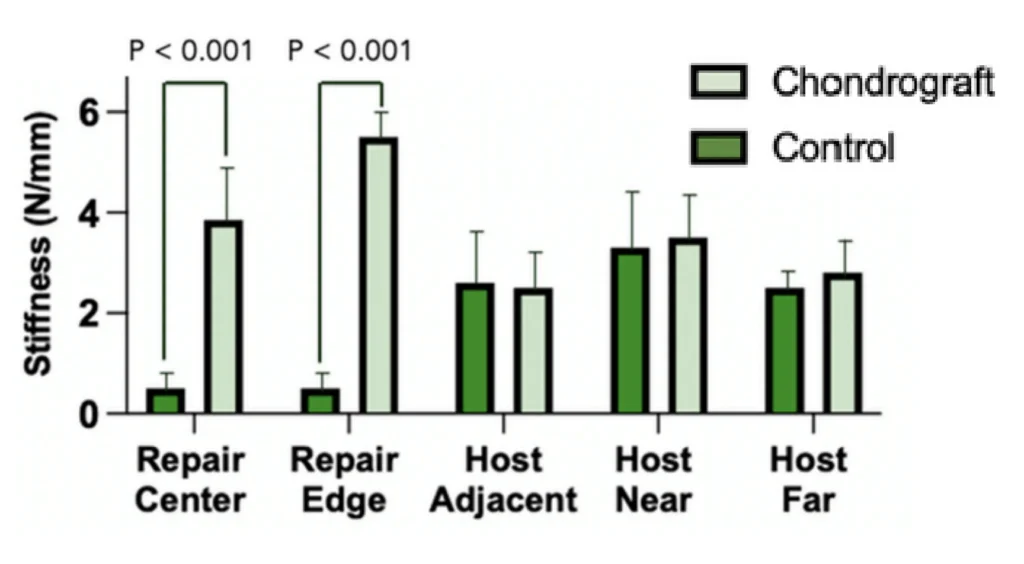
Figure 5. Histology of Chondrograft™ and control defects at 52-weeks. Arrows represent defect margins, * represents the PLLA pin tract.
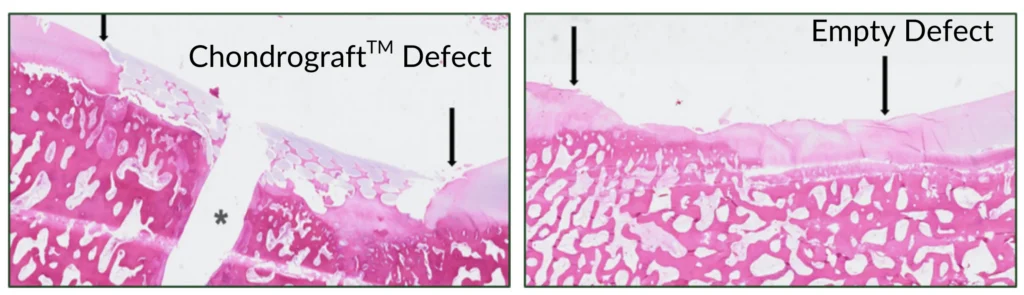
Figure 6. Chondrograft™ results in excellent defect filling and integration to the surrounding cartilage.